
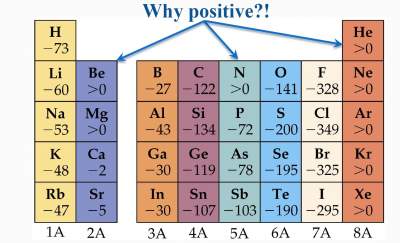
Uncertainty in the final data figures is given in parentheses. The most electronegative element known is fluorine while the element having the highest electron affinity is chlorine.TABLE 4.4 Electron Affinities of Atoms, Molecules, and RadicalsĮlectron affinity of an atom (molecule or radical) is defined as the energy difference between the lowest (ground) state of the neutral and the lowest state of the corresponding negative ion in the gas phase.ĭata are limited to those negative ions which, by virtue of their positive electron affinity, are stable. On the other hand, electron affinity is known to be fixed since the electron releases almost a similar amount of energy when it is added to an atom. The value of electronegativity generally lies between 0.7 to 3.98. However, electron affinity is higher if the nuclear charge of the element is higher. The greater the energy released in the process of taking up the extra electron, the greater will be the electron affinity. This is because energy is usually released in this process, as opposed to ionization energy, which requires energy. Notice the sign on the energy is negative. Atom(g) + Electron(g) Negative ion(g) + Energy. Electron Affinity is the energy associated with the addition of an electon to a gaseous atom. When an element tends to exhibit a stronger attracting ability, the electronegativity of that element is higher. The electron affinity of an element may be defined as amount of energy which is released when an extra electron enters the valence orbital of an isolated neutral atom to form a negative ion. Electron affinity, on the other hand, is measured in kJ/mol. On the other hand, electron affinity is related to the release of energy when an electron gets added to an atom.Įlectronegativity is qualitative while electron affinity is quantitative.Įlectronegativity is generally a unitless quantity but is defined in regards to Pauling. The property due to which the electron gets attracted to the atom is known as electronegativity. The element having the highest electron affinity is Chlorine and the element having the lowest electron affinity is Neon.Īns: Given below are some of the key differences between electronegativity and electron affinity. The most electronegative element known is Fluorine whereas, on the other hand, the least electronegative element known is Francium. The maximum value of electron affinity is when the nuclear charge is more. In chemistry and atomic physics, the electron affinity of an atom or molecule is defined as: the change in energy (in kJ/mole) of a neutral atom or molecule (in the gaseous phase) when an electron is added to the atom to form a negative ion. The maximum value of electronegativity is when the attracting force is high. It is associated with the atom present in a molecule. Ionization Energy and Electron Affinity Section 7.4-7. The electron affinity is a measure of the electron attracting ability. X (g) + 1e X - (g) The 1st electron affinity is usually exothermic (in some cases it is zero). It is either applied to an atom or a molecule. The electron affinity is defined as the energy change when 1 mole of gaseous negative ions is formed from 1 mole of gaseous atoms and 1 mole of electrons. Given below is the table that consists of the detailed differences of both the terms.ĭifference Between Electronegativity and Electron AffinityĮlectronegativity refers to the ability of the atoms to attract the electrons from the other elements.Įlectron affinity refers to the amount of energy that is liberated whenever a molecule or a neutral atom tends to acquire an electron from the other elements. Understanding the differences between electronegativity and electron affinity is important so that we do not use them interchangeably.
In this article, we will study about electronegativity vs electron affinity in detail. However, electronegativity is the result of the property of attraction, while electron affinity is the result of the change in energy. Both electronegativity and electron affinity deal with the movement of the electrons. These two properties are often known to be interrelated but they are not interchangeable. On the other hand, electron affinity is related to the release of the energy whenever an electron tends to get added to an atom. The primary difference between electronegativity and electron affinity lies in the fact that electronegativity is the property related to the ability of an electron to attract towards an atom. Electron affinity Definition: The energy released when an electron is added to a gaseous atom which is in its ground state to form a gaseous negative ion is defined as the first electron affinity. Both electronegativity and electron affinity are two different chemical properties that are associated with the elements. Electron affinity, Electronegativity, Ionization energy 1.
